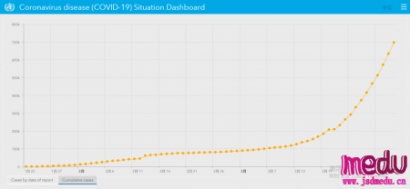
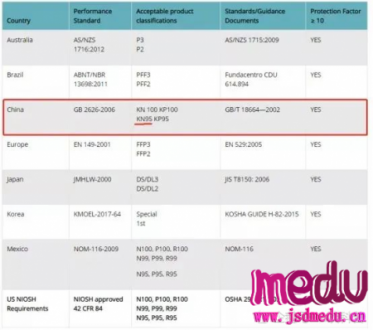
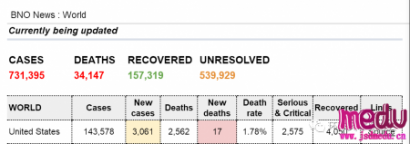
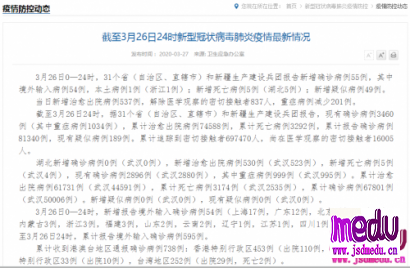
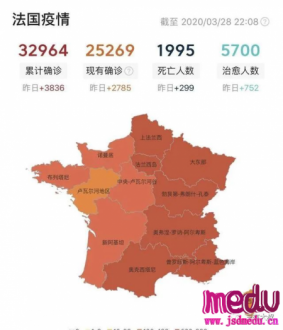
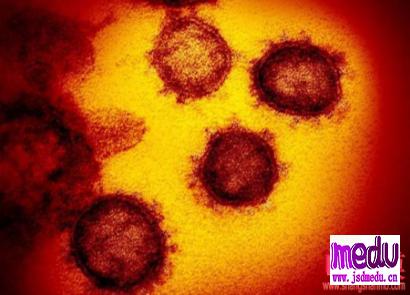
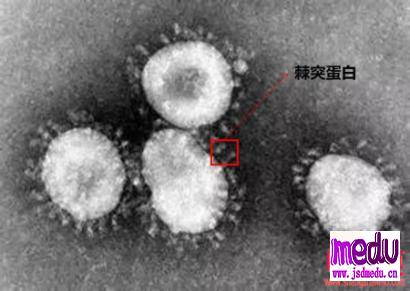
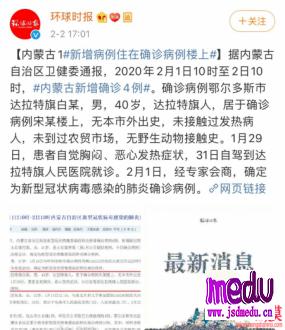
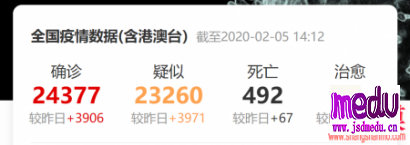
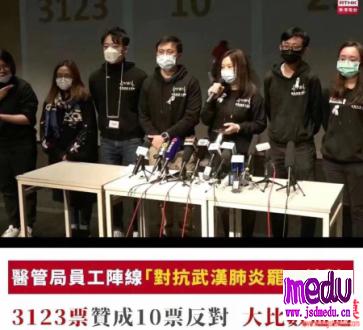
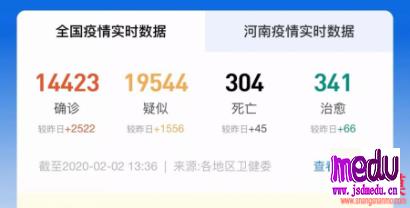
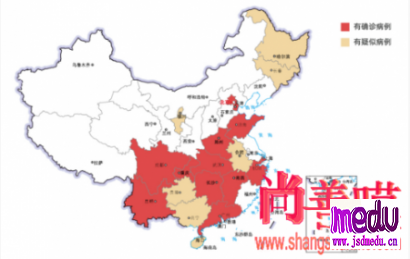
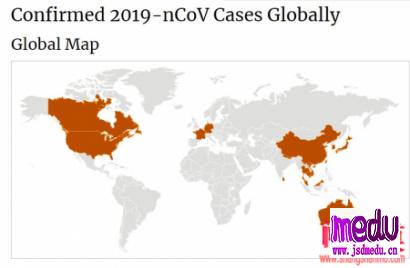
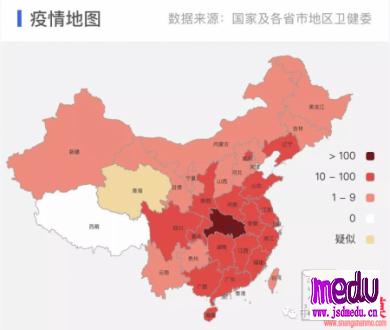
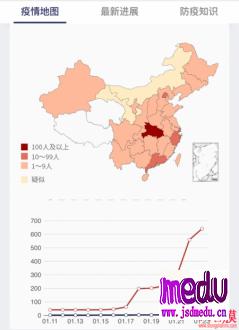
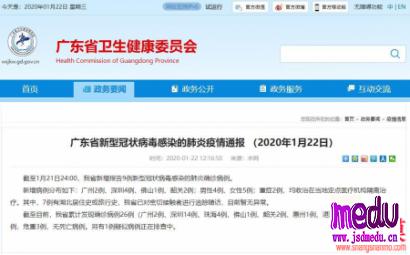
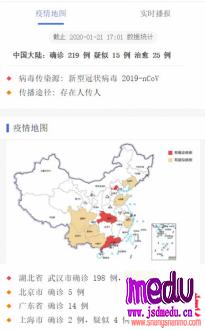
TheChina-JapanFriendshipHospitalinBeijingwillbeginclinicaltrialson270mildlyandmoderatelyillpatientsinfectedwiththenovelcoronavirususinganexperimentaldrugfromtheUnitedStatesclaimedtohavesuccessfullytreatedacaseintheUS.
ThePhaseIIIclinicaltrialwillrunfromFeb3toApril27.ItisadministeredbydoctorCaoBinfromtheBeijinghospitalandthetrialwillbecarriedoutinWuhan,Hubeiprovince,epicenteroftheoutbreak,thehospitalsaidinanonlinestatementonSunday.

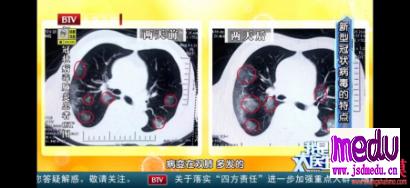
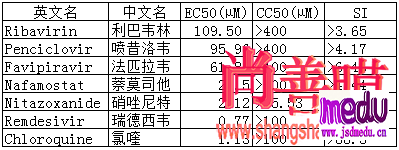
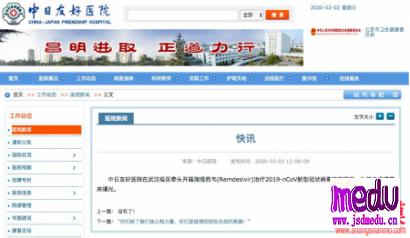
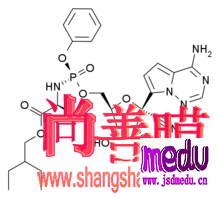
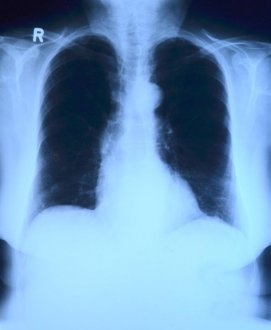
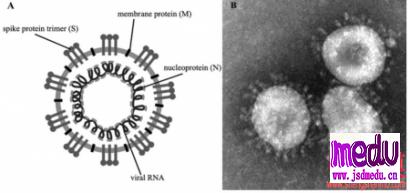
Thedrugintrialiscalledremdesivir,developedbyUSbiotechcompanyGileadSciences.ThemedicinewasgiventothefirstUScase,a35-yearoldmanwhotestedpositiveforthevirus,whosepneumoniasymptomsappearedtoimprovewithinadaywithnoobvioussideeffectsafterthedrugwasadministered,accordingtoapaperpublishedintheNewEnglandJournalofMedicinelastweek.

ThedrugmakersaidinanonlinestatementonFridaythedrugis"notyetlicensedorapprovedanywheregloballyandhasnotbeendemonstratedtobesafeoreffectiveforanyuse".However,thedrugwasapprovedforuseoncompassionategroundsandtherequestofphysicians,itadded.
Atthesametime,thecompanysaiditisworkingwithhealthauthoritiesinChinatoestablishaclinicaltrialtodeterminewhetherremdesivircansafelyandeffectivelybeusedtotreatthecoronavirus.

TheCenterforDrugEvaluationoftheNationalMedicalProductsAdministrationinChinahasreceivedandapprovedtheapplicationonSundaytocarryouttheexperiment,accordingtoitsofficialwebsite.